Annato plant, a source of tocopherol-free tocotrienol.
When I lost my short-term memory six weeks after starting Lipitor, I immediately suspected my new medicine and discontinued the drug. At my next NASA physical, I was urged to resume taking it and reluctantly agreed, only to suffer a much worse memory loss a few weeks later. For 12 hours, I suddenly became a teenager with total recall for my high school days, but absolutely no awareness that I was a doctor, married with children and a former NASA astronaut. This began my decade of research on the subject.
To my amazement, I discovered an extraordinary lack of information in my own medical community as to the true spectrum of statin drug side effects. I started a Web site (www.spacedoc.com), which has become a major message board and resource for those with statin drug-associated side effects. My four books—Lipitor, Thief of Memory, Statin Drug Side Effects, Statin Damage Crisis and The Dark Side of Statins—present the reasons for adverse effects of statins on the human body.
Generally speaking, the spectrum of statin drug damages include cognitive dysfunction, emotional and behavioral changes, myopathy, neuropathy and a variety of chronic neuromuscular degenerative conditions as well as an ALS-like syndrome. Transient global amnesia, a once rare condition prior to statin drug use, is but the tip of the iceberg of the many other cognitive complaints: increased forgetfulness, disorientation, confusion and increased severity of pre-existing senility resulting from excessive inhibition of cholesterol bioavailability by the increasingly powerful statins. Research has revealed that our cholesterol levels are less relevant indicators of atherosclerosis and increased cardiovascular risk, while inflammation is known to be a fundamental cause.
Newer studies show that statins work in lowering cardiovascular risk not only because of their cholesterol manipulation, but also because they are powerful inhibitors of inflammation—impeding nuclear factor kappa-B, a transcriptase factor essential to our entire immuno-defense system (1). After 35 years of merciless cholesterol bashing by national health leadership, recent clinical trials (including the Jupiter study) firmly document the decreased importance of cholesterol to the atherosclerotic process, citing LDL as a poor indicator for cardiovascular risk (2).
While studying and writing as much as I could about statin side effects, I also set out on my decade-long search for natural alternatives to statin’s dark side. Of the many products I found beneficial, a lesser-known vitamin E—tocotrienol—has become a rising star.
Tocotrienols, with their anti-inflammatory and enhanced antioxidation effect could not have come at a better time for the tens of thousands of people whose muscles and nerves have been inflamed by the use of statins. Many have been incapacitated, myself included. There is now a premium on natural biochemicals that have both anti-inflammatory as well as antioxidant actions. Tocotrienols are a superior candidate worthy of further consideration, and for these particular applications should replace the now redundant old vitamin E guy, alpha-tocopherol, that was around when I was in medical school nearly 60 years ago. My hat goes off to Dr. Barrie Tan, founder of American River Nutrition, Inc., who discovered the annatto source of tocotrienols. Annatto tocotrienol is naturally tocopherol-free and only contains delta- and gamma-tocotrienols, the vitamin E compounds most potent in anti-inflammation, antioxidation and cholesterol management.
What Is Tocotrienol and Why Does It Work So Well?
Tocotrienol is one of the two branches of the vitamin E family, the other one being the more famous tocopherols. In simple terms, nature makes four tocopherols and four tocotrienols, each designated with the Greek symbols alpha, beta, gamma and delta. Alpha-tocopherol is the most well-known form, and has been popularized since its discovery in 1922. Tocotrienols, on the other hand, are almost unknown. Too much emphasis has been placed on the functions of vitamin E tocopherol, and the science of tocotrienol is only now emerging, warranting the publication of a first-ever tocotrienol book, Tocotrienols: Vitamin E Beyond Tocopherols, in 2008.
As vitamin E molecules, both tocopherols and tocotrienols have similar chemical entities. So, why does tocotrienol work so differently from tocopherol? The answer is found in a detailed look at the subtle differences in molecular structure.
The vitamin E molecule looks like a tadpole, with a head and a tail.
The head of vitamin E performs the antioxidant work, and determines whether a vitamin E is designated alpha, beta, gamma or delta depending on the amount of substituted methyl groups. Generally, less methyl groups (desmethyl) signify smaller molecular size and greater potency.
The tail anchors into lipid membranes constituting the cell wall and are abundant in all cells of the body.
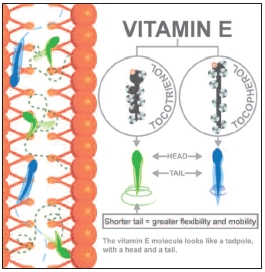 Because the membrane is made of fatty acids and oftentimes unsaturated fats such as the beneficial omega-3s (as found in the eye and brain, for example), cell walls of unstable lipids need a lot of protection from oxidation. Within these membranes is a great space for vitamin E to be. They police radical oxidants to prevent cell membrane damages.
Because the membrane is made of fatty acids and oftentimes unsaturated fats such as the beneficial omega-3s (as found in the eye and brain, for example), cell walls of unstable lipids need a lot of protection from oxidation. Within these membranes is a great space for vitamin E to be. They police radical oxidants to prevent cell membrane damages.
While both tocopherols and tocotrienols have the same structural head, tocotrienols have a shorter and more flexible tail, enabling them to move freely through the cell membrane to cover larger areas for added protection. The smaller tocotrienol tadpole with its shorter tail zigzags through the cell membrane faster and with greater agility, while the larger tocopherol tadpole with its longer tail staggers through the membrane more slowly and without cellular suppleness.
Tocotrienols vs. Statins
Like statins, tocotrienols impact the HMGR pathway (3-hydroxy-3-methyl-glutaryl-CoA reductase, the enzyme/protein responsible for the body’s cholesterol production). Statins are competitive inhibitors of the enzyme and block not only cholesterol, but also other important intermediates in the pathway. This is the culprit causing the various undesired side effects that I have coined the “Statin Damage Crisis.”
Unlike statins, tocotrienols “dial down” cholesterol production without side effects, and hence they are discriminating cholesterol reducers. They also degrade the HMGR protein, a mechanism of action that was elucidated in the early 1990s (3), and revalidated 15 years later (4). In both earlier and recent studies, only desmethyl tocotrienols (namely, delta- and gamma- isomers) among the E vitamins had this effect on HMGR.
In the various clinical studies administering tocotrienol supplements, patients did not experience side effects as are observed with statin cholesterol-lowering drugs, many of which are due to statin’s inhibition of CoQ10 synthesis. Instead, preliminary open label studies with 75 mg/day tocotrienols increased CoQ10 levels by up to 20% (5). Tocotrienol’s ability to increase our body’s CoQ10 production has recently been shown scientifically (6–8).
Interestingly, tocotrienols and low-dose statins were shown to work synergistically in clinical studies for cholesterol management (9) and animal studies for cancer treatment and prevention (10).
While statins lower LDL cholesterol by a startling 40–60%, tocotrienols have been shown to lower these levels by a modest 15–20%, but without side effects. Tocotrienol’s synergism with statins may advocate taking the two in combination, especially in an effort to lower statin dosage for a patient population with severe side effects.
Addressing the Importance of Inflammation as Cardiovascular Risk Factor
Following the Jupiter and various other studies, it is now clear that high cholesterol should not be singled out as a risk factor for cardiovascular disease. In fact, approximately half of the patients with cardiac events have perfectly “normal” cholesterol levels, but may be suffering from undiagnosed inflammation that contributes to oxidative and atherosclerotic processes. Many may not realize that statins’ anti-inflammatory properties first emerged with evidence of their lowering CRP levels and cardiac events in a group of healthy men and women boasting normal cholesterol levels (2).

|
| Duane Graveline, M.D., MPH, is a Vermont family doctor, aerospace medical research scientist, USAF flight surgeon and NASA astronaut. He now resides in Merritt Island, FL and is still affiliated with the space program as consultant to the special cosmic radiation hazards unit and NASA’s “return to the moon and on to Mars” program. |

|
Like statins, tocotrienols were shown to have potent anti-inflammatory properties minus the side effects. Qureshi and others evaluated the effect of tocotrienols in reducing inflammation in experimental mice (11). They demonstrated that alpha-, gamma- and delta-tocotrienols strongly inhibited the inflammatory response using such markers as chymotrypsin, trypsin and tumor necrosis factor-a, with delta-
tocotrienol being the most effective. The results of this study demonstrated that the use of tocotrienols can function as a powerful proteasome modulator, while increasing the immune system’s ability to fight inflammation. At the same time, tocotrienols induce a hormone that produces an anti-inflammatory steroid to block inflammation directly.
In another study, the same lab group showed that delta-tocotrienol efficiently reduced nitric oxide and tumor necrosis factor-a levels in chickens (12), and that when combined with natural alternatives quercetin (a polyphenol) or riboflavin (a B vitamin), the two worked in synergism. Delta-tocotrienol was also the most potent vitamin E compound in the downregulation of COX-2 expression without affecting the housekeeping COX-1enzyme, and potently reduced major proinflammatory cytokine IL-6 levels (13, 14).
Winning the Fight
As awareness of statin side effects grows, consumers increasingly turn to natural nutritional alternatives to address preventive health measures. Although less dramatic than statins, research on tocotrienols shows them to be effective in cholesterol management and anti-inflammation without the sometimes debilitating side effects. In addition, with their synergism established, tocotrienols may allow patients to reduce statin dosages, especially when they are experiencing severe side effects. In this sense, tocotrienols are taking a stand in winning the fight against the statin damage crisis. WF
References
1. G.J. Blake and P.M. Ridker, “Are Statins Anti-Inflammatory?” Curr. Control. Trials Cardiovasc. Med. 1 (3), 161–165 (2000).
2. P.M., Ridker, et al., “Rosuvastatin to Prevent Vascular Events In Men And Women With Elevated C-Reactive Protein,” N. Engl. J. Med. 359 (21), 2195–2207 (2008).
3. B.C., Pearce, et al., “Hypocholesterolemic Activity of Synthetic and Natural Tocotrienols,” J. Med. Chem. 35 (20), 3595–3606 (1992).
4. B.L. Song and R.A. DeBose-Boyd, “Insig-Dependent Ubiquitination and Degradation of 3-Hydroxy-3-Methylglutaryl Coenzyme a Reductase Stimulated by Delta- and Gamma-Tocotrienols,” J. Biol. Chem. 281 (35), 25054–25061 (2006).
5. B. Tan and A.M. Mueller, “Tocotrienols in Cardiometabolic Diseases,” in Tocotrienols: Vitamin E beyond Tocopherol, R. Watson and V. Preedy, Eds. (AOCS/CRC Press, Boca Raton, FL, 2008, p. 257-273).
6. M., Bentinger, et al., “Stimulation of Coenzyme Q Synthesis,” Biofactors, 32 (1–4), 99–111 (2008).
7. V.E., Kagan, J.P. Fabisiak and P.J. Quinn, “Coenzyme Q and Vitamin E Need Each Other as Antioxidants,” Protoplasma, 214:,p. 11–18 (2000).
8. Y., Zhang, M. Turunen and E.L. Appelkvist, “Restricted Uptake of Dietary Coenzyme Q is in Contrast to the Unrestricted Uptake of Alpha-Tocopherol into Rat Organs and Cells,” J. Nutr. 126 (9), 2089–2097 (1996).
9. A.A., Qureshi, et al., “Synergistic Effect of Tocotrienol-Rich Fraction (TRF(25)) of Rice Bran and Lovastatin on Lipid Parameters in Hypercholesterolemic Humans,” J. Nutr. Biochem. 12 (6), 318–329 (2001).
10. J.A., McAnally, et al., “Tocotrienols Potentiate Lovastatin-Mediated Growth Suppression In Vitro and In vivo,” Exp. Biol. Med. 232 (4), 523–531 (2007).
11. A.A., Qureshi, et al., “Tocotrienols Inhibit Lipopolysaccharide-Induced Pro-Inflammatory Cytokines in Macrophages Of Female Mice,” Lipids Health Dis. 9 (1), 143 (2011).
12. A.A., Qureshi, et al., “Delta-Tocotrienol and Quercetin Reduce Serum Levels of Nitric Oxide and Lipid Parameters in Female Chickens,” Lipids Health Dis., 10, 39 (2011).
13. K. Nesaretnam and P. Meganathan, “Tocotrienols: Inflammation and Cancer,” Ann. NY Acad. Sci. 1229 (1), 18–22 (2011).
14. M.L., Yam, et al., “Tocotrienols Suppress Proinflammatory Markers and Cyclooxygenase-2 Expression in RAW264.7 Macrophages,” Lipids, 44 (9), 787–797 (2009).
Published in WholeFoods Magazine, October 2011









