
Quicklinks
WholeFoods Magazine
Supplements' Weakest Link

Any chain is only as strong as its weakest link, and that’s certainly true in the dietary supplements and natural products universe. And when it comes to supply chains specifically, despite movement towards clean and simple labels, dietary supplements is one of most the complex.
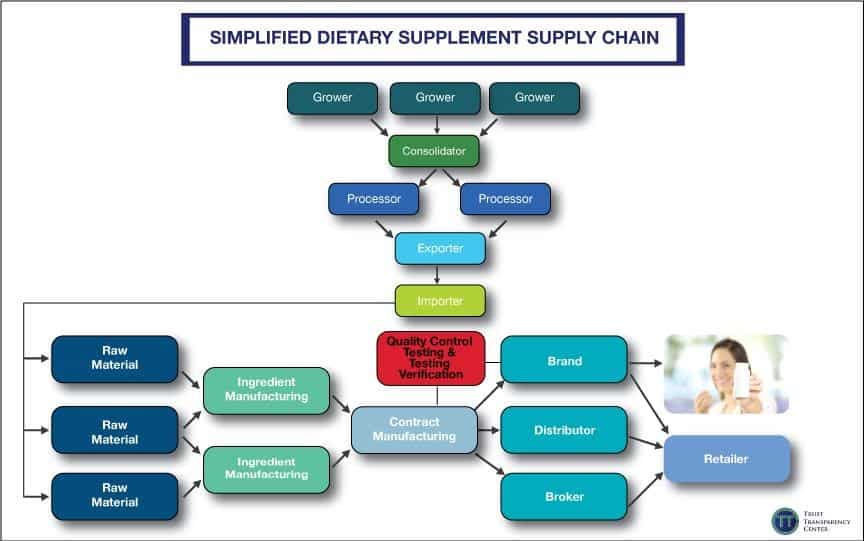 Many of the brands stocked in store or purchased online focus heavily on marketing and product positioning, leaving critical operations such as actual manufacturing and testing to so-called “expert” outsourced companies. Emerging brands especially will make use of these services, exploiting low minimum commitments rather than investing heavily in their own capabilities. While this is easily understood at operational and business levels, it opens the door to corner- and cost-cutting and other unsavory business practices. In fact, all too often it becomes a race to the bottom as margins erode, driving additional cost-cutting in a vicious cycle.
Many of the brands stocked in store or purchased online focus heavily on marketing and product positioning, leaving critical operations such as actual manufacturing and testing to so-called “expert” outsourced companies. Emerging brands especially will make use of these services, exploiting low minimum commitments rather than investing heavily in their own capabilities. While this is easily understood at operational and business levels, it opens the door to corner- and cost-cutting and other unsavory business practices. In fact, all too often it becomes a race to the bottom as margins erode, driving additional cost-cutting in a vicious cycle.
Let’s be clear: There are world-class operations making some of our dietary supplements and natural products. These organizations focus on quality, have long-term relationships with research-oriented ingredient companies, and they actively protect and build supply chain into a strategic advantage. They are industry-savvy and are able to bring reams of institutional knowledge to their client base. They are active within the industry and they most often are not the cheapest source.
To what extent is this exposure at contract manufacturing an industry risk?
The recent ABH Pharma recall perfectly illustrates this vulnerability. We have an agency, the FDA, charged with enforcing existing regulations and GMPs that has negligently allowed this company to operate for years, putting numerous companies and products at risk. At the same time, the market, economics, human behavior, and other factors, have allowed companies like ABH to exist and even thrive.
It is extremely frustrating to witness industry self-policing efforts fail to make maximum impact in this critical area of our supply chains. One such self-policing example is the American Botanical Council’s Botanical Adulterants Prevention Program (BAPP) which has now published 20 bulletins describing at-risk botanicals and how to properly test for common adulterants. While some manufacturers quickly incorporate these bulletins into their quality control procedures, others remain deliberately or naively unaware. Trust Transparency Center has directly interacted with several contract manufacturers that really should know better, as their client list includes some pretty large organizations with loads at stake.
This situation provides the retailer as gatekeeper a critical role, and even more importantly, opportunity to add value, cement trust and differentiate. The ABH recall implicated 859 brands/products. It raises questions about these products certainly, and in the bigger picture speaks to both product and brand vetting, as well as everyone’s ability to drive transparency and accountability in our industry.
Just how are products vetted? Do we know where/how they’re manufactured? And if companies fail to disclose this information, do we want to even work with them? Are any of the ABH 859 on shelf? Do companies test their finished products at all? Can we leverage positively those that do? Can we absolutely demand objective third party testing and expect to see the evidence?
If it’s true that our industry is one major recall away from losing that fragile credibility that drives our businesses, can’t we all start asking better questions and demanding the practices we support be incorporated and actively demonstrated by the brands we deal with?
There’s a lot at stake.
 Many of the brands stocked in store or purchased online focus heavily on marketing and product positioning, leaving critical operations such as actual manufacturing and testing to so-called “expert” outsourced companies. Emerging brands especially will make use of these services, exploiting low minimum commitments rather than investing heavily in their own capabilities. While this is easily understood at operational and business levels, it opens the door to corner- and cost-cutting and other unsavory business practices. In fact, all too often it becomes a race to the bottom as margins erode, driving additional cost-cutting in a vicious cycle.
Many of the brands stocked in store or purchased online focus heavily on marketing and product positioning, leaving critical operations such as actual manufacturing and testing to so-called “expert” outsourced companies. Emerging brands especially will make use of these services, exploiting low minimum commitments rather than investing heavily in their own capabilities. While this is easily understood at operational and business levels, it opens the door to corner- and cost-cutting and other unsavory business practices. In fact, all too often it becomes a race to the bottom as margins erode, driving additional cost-cutting in a vicious cycle.Let’s be clear: There are world-class operations making some of our dietary supplements and natural products. These organizations focus on quality, have long-term relationships with research-oriented ingredient companies, and they actively protect and build supply chain into a strategic advantage. They are industry-savvy and are able to bring reams of institutional knowledge to their client base. They are active within the industry and they most often are not the cheapest source.
Related: The Evolution of Contract Manufacturing Contract Manufacturer Issues Major Recall of Dietary Supplements Contract Manufacturers ID 10 Top Trends for 2020
It has been estimated that less than 15% of the supplement contract manufacturers in the U.S. belong to trade associations (Source: Trust Transparency Center database). While many of these manufacturers can and do claim a GMP certification or audit, there is a wide range of expertise and quality, and so much incentive to take shortcuts. Newer market entries and emerging brands are at the most risk of being exploited by unscrupulous contractors. These organizations are often reverse-engineering or knocking off basic products, which means that the most aggressive pre-produce in bulk to create cost efficiency, then are ready to slap any label on the goods they sell to ignorant buyers. There is no expiry date in sight, no stability testing, and limited, if any, finished product testing. Third-party product testing and stability are certainly not in play, cost cutting has eliminated science-backed ingredients in favor of non-substantiated commodities and knockoffs, and active dosages drop in favor of “fairy-dusting.” On top of these issues, many newer brands, especially online only, have no concept of their obligations under GMP, instead relying on their manufacturers to cover them. The contractors, though, operate lean shops, have short-changed many QA and QC processes, and many of them in fact are actually a GMP liability. This is a huge industry issue, that has been newly popularized with low barriers to entry.To what extent is this exposure at contract manufacturing an industry risk?
The recent ABH Pharma recall perfectly illustrates this vulnerability. We have an agency, the FDA, charged with enforcing existing regulations and GMPs that has negligently allowed this company to operate for years, putting numerous companies and products at risk. At the same time, the market, economics, human behavior, and other factors, have allowed companies like ABH to exist and even thrive.
It is extremely frustrating to witness industry self-policing efforts fail to make maximum impact in this critical area of our supply chains. One such self-policing example is the American Botanical Council’s Botanical Adulterants Prevention Program (BAPP) which has now published 20 bulletins describing at-risk botanicals and how to properly test for common adulterants. While some manufacturers quickly incorporate these bulletins into their quality control procedures, others remain deliberately or naively unaware. Trust Transparency Center has directly interacted with several contract manufacturers that really should know better, as their client list includes some pretty large organizations with loads at stake.
This situation provides the retailer as gatekeeper a critical role, and even more importantly, opportunity to add value, cement trust and differentiate. The ABH recall implicated 859 brands/products. It raises questions about these products certainly, and in the bigger picture speaks to both product and brand vetting, as well as everyone’s ability to drive transparency and accountability in our industry.
Just how are products vetted? Do we know where/how they’re manufactured? And if companies fail to disclose this information, do we want to even work with them? Are any of the ABH 859 on shelf? Do companies test their finished products at all? Can we leverage positively those that do? Can we absolutely demand objective third party testing and expect to see the evidence?
If it’s true that our industry is one major recall away from losing that fragile credibility that drives our businesses, can’t we all start asking better questions and demanding the practices we support be incorporated and actively demonstrated by the brands we deal with?
There’s a lot at stake.







