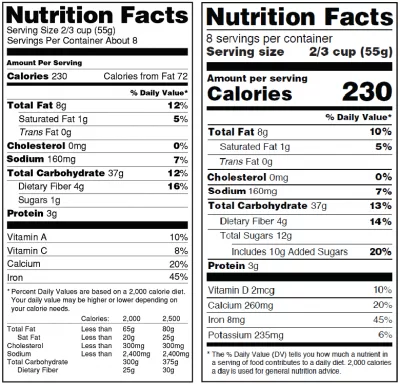
The new labels, first announced in 2016, list calories in a larger font, serving sizes that are more realistic to what Americans eat on average, and have a new space on the label for added sugars.

While the old label lists total grams of sugars, it doesn't distinguish between sugars that are naturally occurring in foods like fruits and vegetables, and sugars that meet the definition of added sugars. The new label now includes added sugars in grams and as percent Daily Value. “Added sugars” are regarded as worse than sugars occurring naturally in, say, fruit.
"Our update to the iconic Nutrition Facts label includes significant changes to help consumers make more informed dietary choices, and we are already seeing the new label on many products," Gottlieb says in the press statement.
The guidance for added sugars specifically relates to honey, maple syrup and certain cranberry products. While honey and maple syrup meet the definition of added sugars, there were concerns from industry leaders that declaring added sugars on their single-ingredient products may lead to consumer confusion. Cranberry juice manufacturers added that their products need to be sweetened for palatability. The new solution is to give manufacturers a symbol to put on packaging immediately after the added sugars daily value, directing consumers to language that provides truthful and contextual information about the “added” sugars in their products, according to the FDA.
Data shows it's difficult to meet nutrient needs while staying within calorie limits if you consume more than 10% of your total daily calories from added sugar, and this is consistent with the 2015-2020 Dietary Guidelines for Americans.
Other changes include updating the list of nutrients on labels. Vitamin D and potassium will be required on the label. Calcium and iron will continue to be required. Vitamins A and C will no longer be required but can be included on a voluntary basis.
And while continuing to require “Total Fat,” “Saturated Fat,” and “Trans Fat” on the label, “Calories from Fat” is being removed because research shows the type of fat is more important than the amount, according to the FDA statement.
Introduced during the Obama administration, last year President Trump’s administration announced label changes were delayed “indefinitely.” The announcement of these guidelines, which includes a confirmation of the 2020 deadline, shows that while the FDA is committed to giving nutrition labels an overhaul, they are also listening to the concerns of the food industry.
The FDA will embark on an educational campaign for consumers when the changes go into effect in 2020 and 2021. The earlier compliance date is for manufacturers with annual sales of more than $10 million; the later one is for those with annual sales below that amount.
The FDA had initially suggested a deadline of July 26, 2018, for implementing the new label, but last June announced it had decided to delay that until 2020 and 2021 because some manufacturers and trade associations were concerned about doing it before the mandatory GMO ingredient labeling was required.
See related articles: